What is the structure of cyano compound with 6 carbon atom and their IUPAC naming?
Cyano compounds
are those compounds in which alkyl groups are attached with the carbon atom of
– CN functional group. Cyano compounds are also known as nitrile compounds.
The general
formula of cyano compound is CnH2n+1–CN. Depending on
the number of cyano group, cyano compounds may be mono cyano,dicyano,tricyano
etc.
In case of IUPAC
nomenclature of cyano compounds, the carbon atom of cyano group must be
considered. That is, we select long chain including cyano group and then
numbering the carbon atom.
According to
IUPAC rules, the cyano compounds are named as,
Root alkane +
nitrile = alkane nitrile
The carbon atom
of root alkene must be equal to the carbon atom present in the long chain of
the concern cyano compound.
For example,
butane nitrile, pentane nitrile, hexane nitrile etc.
If the cyano compounds
contain branch chain,then the naming of the branch chain with their position in
the alphabetic order first and then naming of the root chain.
Again, if a
cyano compound contain more than one cyano group, then in nomenclature, the
sequence is, position of the cyano group + root alkane name + number of cyanogroup [ di, tri, tetra etc ] + carbonitrile.
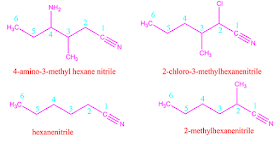
For example,4-amino 3-methyl hexane nitrile.
The structure of
some cyano compounds with 6 carbon atom and their IUPAC nomenclature are shown
below.
Summary
What is cyano compounds in chemistry ?
What is structure and nomenclature of cyano compound with 6-carbon atom ?



No comments:
Post a Comment